For the technically interested reader, what follows on this page is a description of the S 433 amino acid analyzer explaining the different building blocks according to the flow-diagram. The chromatographic claim of high resolution and stable low noise baselines combined with higher sensitivity and a larger measuring range – compared to other classical amino acid analyzers – are substantiated along the line.
The Sykam S 433 amino acid analyzer is the culmination of over 30 years of experience with the production and development of classical amino acid analyzers at Sykam GmbH in Germany. The S 433 together with a range of ready to use columns, buffers and reagents are a complete and reliable solution aimed at daily routine analysis of physiological fluids, food and feed hydrolysates, infusion solutions and other sample matrixes like for instance fermenter-broths.
The flow-diagram below differentiates between four building blocks:
- The refrigerated buffer/reagent organiser unit
- The combined buffer selection- and gradiënt mixing valves and buffer pump unit
- The autosampler
- The combined reagent pump, column temperature control, post-column reactor and detector unit
Each described in detail on this page.
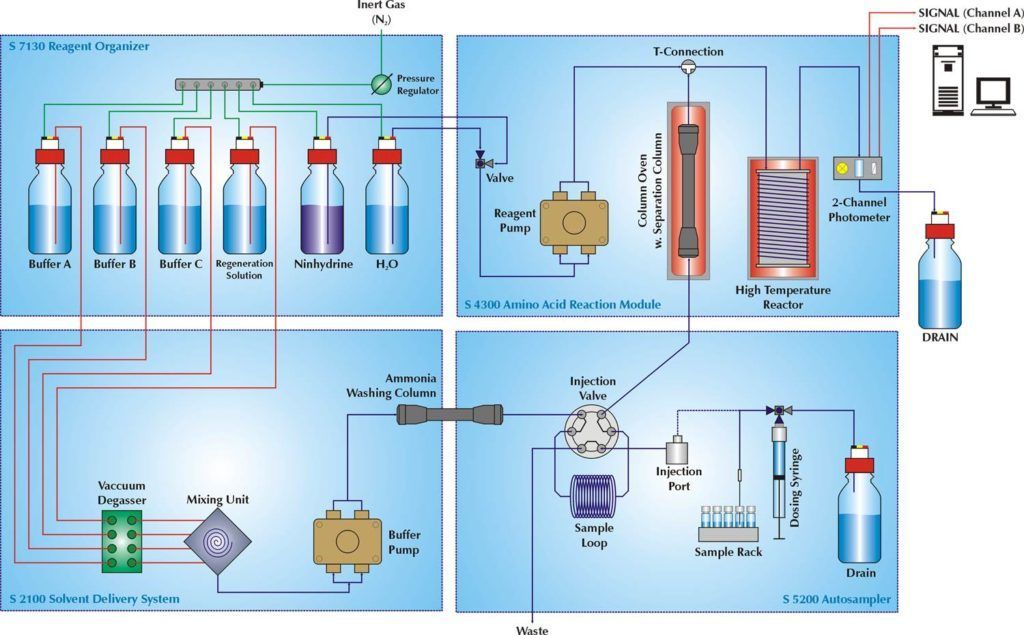


The reagent organizer contains the buffers, the reactor washing solution and Ninhydrin reagent. The reagent organizer is refrigerated and all bottles are placed under inert-gas (Helium or Nitrogen) to ensure maximum reagent stability. All bottle caps are equiped with closing valves for the liquid and inert-gas lines. The reagent bottle has an additonal inert-gas purging line (a gas-tubing with purging filter positioned at the bottom of the reagent bottle) to expel any residual oxigen. This eliminates the need for any external degassing or mixing the reagent outside of the instrument.

The Ninhydrin reagent is supplied in one liter bottles accompanied by a syringe with two milliliters of catalyst. Just pouring the Ninhydrin and adding the 2 ml of catalyst-solution following a short period helium or nitrogen purging on the instrument is all that is needed to obtain a stable reagent on the analyzer.


The buffer pump unit is based on a special version of the S 2100 HPLC pump as described in the HPLC pages of this website. It contains the buffer selection valves which operate in gradiënt mode. A built-in vacuum degasserremoves residual gasses from the buffers before reaching the buffer selection valves, gradiënt mixer and pumphead. Active vacuum degassing before the buffer selection valves – instead of a commonly used bubble-trap after the buffer selection valves – is a first step ensuring a stable flow for maintaining a stable baseline and eliminating additional dead-volume which would otherwise result in noticable longer regeneration/equilibration time of the column after the last amino acid elutes. Therefore noting injection to injection time with amino acid analyzers is more interesting that just looking at retention time of the amino acid last eluted. Especially since some analyzers need long time before column regeneration and equilibration is complete.
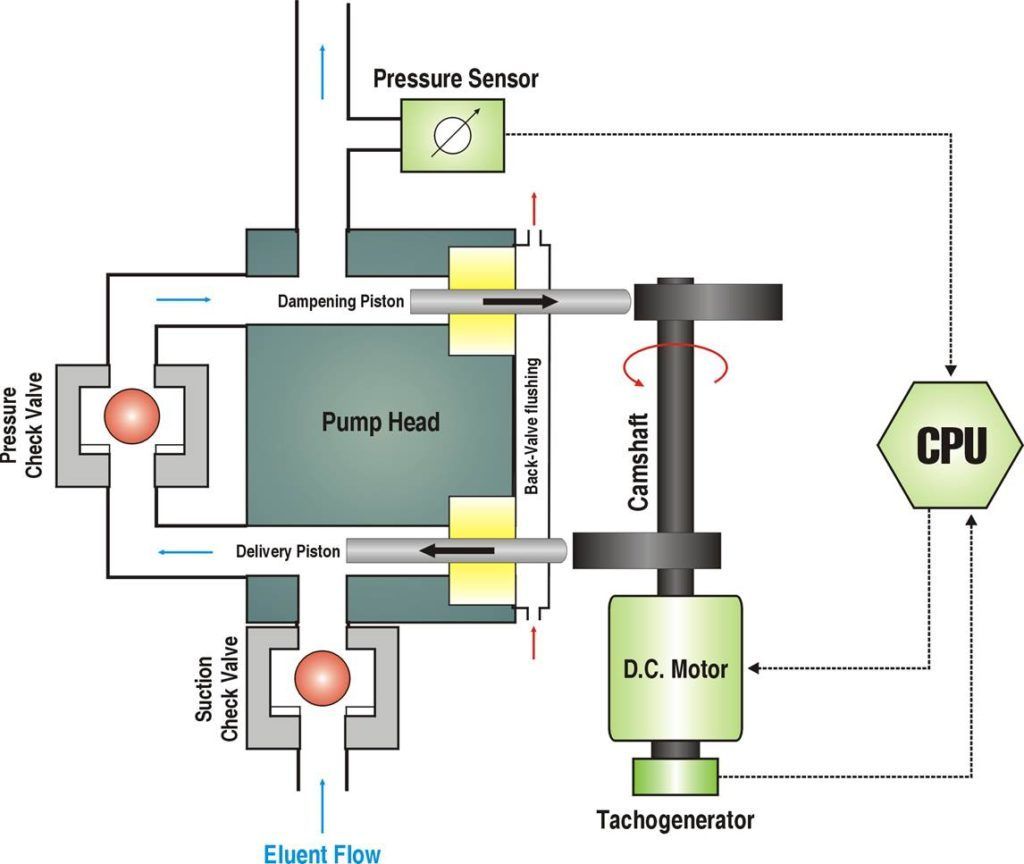
The buffer pump consists of a dual plunger pump-head made of PEEK with ceramic plungers and a back-seal flushing compartment. To ensure a stable pulsation free flow the pump is microprocessor controlled and monitors the feedback of the actual pump displacement volume by an optical tachogenerator and the feedback from the pressure sensor to compensate for any residual pulsation. Active elimination of flow pulsation is the second step of stabilizing the baseline signal.


S 5200 Sample Injector
The autosampler of the S 433 amino acid analyzer is based on the bio-inert PEEK version of the S 5200 autosampler as described in the HPLC section of this website. The autosampler has a variable volume injection with a programmable range of 1 µl up to 100 µl and uses standard 12 x 32 mm vials.
Sample temperature control (+4 oC to +70 oC) is optional and usually not necessary for hydrolysate analysis. The two sample trays of 60 positions each, can be taken from the autosampler – literally – with only one hand, for easy vial loading on the lab-bench.



The Reagent Pump module contains, apart from the pump, the column-compartment (accessible from te side), the post-column reactor and the detector as well. The unit is positioned above the buffer pump.
The Reagent Pump, has the same flow control features as the buffer pump ensuring maximum flow stability. One remarkable difference to the buffer pump, and at the same time a third step contributing to baseline stability, is the fact that the reagent pump is a micro-version pump (0.01 – 2.00 ml/min), whereas the buffer pump is an analytical pump with a flow range of 0.01 to 10 ml/min. When using the same pump for buffer and reagent without flow feedback control features, as most amino analyzers do, one can observe when both pumps are running in-phase – and this happens periodically even when the pumps have different flowrates – that baseline noise increases! Digital smoothing and low data sampling rates or an increased detector time-constant are often used to disguise this effect! With the S 433 amino acid analyzer the buffer and reagent pump never run in-phase, hence lowering baseline noise and pulsation effects in the first place! Assuring asynchronous behavior between the reagent and bufferpump is the third step in stabilizing the baseline!
The Reagent pump has a selection valve to switch between Ninhydrin and washing solution. Apart from – end of sequence – reactor and cuvette washing routines, the reagent pump is programmed to pump washing solution through the reactor during regeneration and equilibration of the column during each run. Using this strategy keeps the reactor and cuvette clean from reagent residues ensuring long term baseline stability and avoiding the need for regular cuvette cleaning as often observed with other amino acid analyzers. The long term baseline stability is proven by the fact that the detector auto-zero function is switched off during normal operation! Sinse the Ninhydrin reagent by itself, is a reactive dye poluting the reactor and detector cuvette over time, using the autozero function of the detector at each run – as is usual with most analyzers – can compromise the detector linearity range by substracting an ever higher background signal without the user noticing the effect! With the Sykam analyzer this effect is simply avoided by the intelligent use of the reagent pump with washing solution. Intelligent programming of washing routines, inlcuding washing of the Ninhydrin pump itself, is the fourth step in maintaining a stable baseline.
The reaction coil is an FEP-tubing coiled around a heater in a heating bath filled with high temperature silicon oil. Each coil is tested according to stringent flow-pressure specifications before it is applied for the S 433 amino acid analyzer. Commonly used PTFE-tubing as a reaction coil has proven to have a shorter long term stability compared to FEP. Upon opening a reactor-chamber containing a PTFE-coil an acidic buffer/reagent smell is often observed and stays unnoticed for some time before causing problems for the analyzer and the analysis. The use of FEP with stringent quality control avoids this problem.
The detector has to comply with some specific demands to ensure a stable baseline and high sensitivity combined with a large as possible measuring range. Low-stray light, low-drift and low noise are features that are nowadays common to most HPLC UV/Vis detectors available on the market. It is however a fact that with an amino acid analyzer one has Ninhydrin reagent with a relatively high viscosity mixing with low viscosity eluents. With measurement at 570 nm and 440 nm one would not expect much trouble, but even the least of pulsation or lack of proper mixing behind the column becomes easily apparent as refractive index effects. Historically this can be illustrated by the fact that an earlier well known analyzer, the “Beckman 6300” used the signal from a third channel at 700 nm – responding only to the RI-effect – as a compensation signal to subtract from the 570 nm and 440 nm channel. Sykam developed the S 433 detector with high quality optics and long-life heavy duty pre-aligned lamps ensuring minimal response to refractive index effects without the need for any digital smoothing, slow response times or third channel baseline subtraction. Pre-alligned lamps and lowest refractive index response of the detector is the fifth step in stabilizing the baseline.
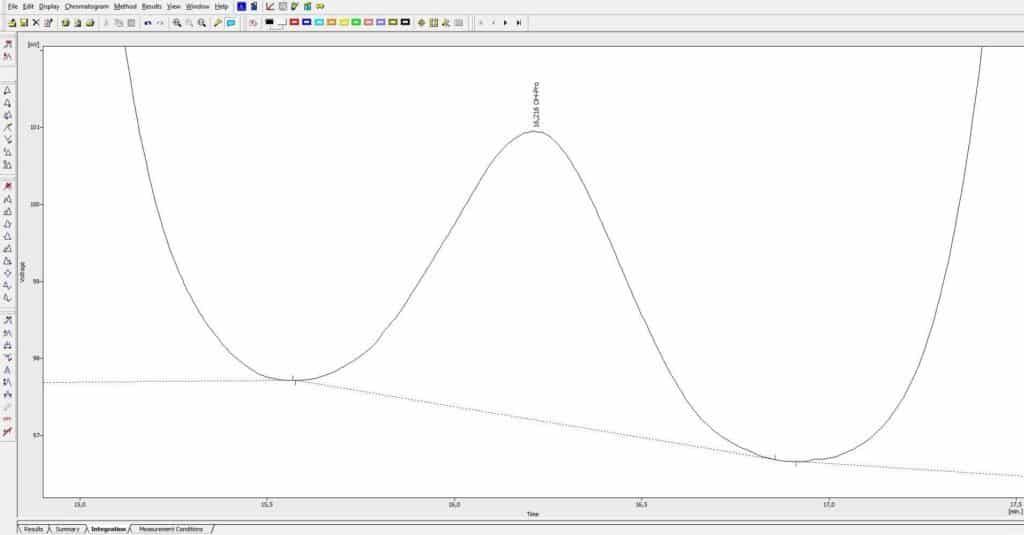
How the above described measures work out on the final analysis result is best illustrated by a practical example that can be easily comparad to other analyzers that you might currently use. Shown here below, is the 3 nmol (amount on column) Hydroxyproline peak shown on the 570 nm channel with a data sampling rate of 6 Hz, hence no digital smoothing, giving an expected low peak-height of 3 mV only and still showing excellent low noise resulting in easy integration! Easy and accurate Integration – of small peaks like often present in chromatograms of physiological fluids from e.g. newborn children – helps ensuring quantitative precision and saves the user lots of time determining the right baseline settings with the often complex chromatograms of these samples. We chose this practical example because one can easily compare this profile to the profile of his/her current amino acid analyzer by running a standard with 3 nmol hydroxyproline (amount on column) and data collection at 6 points per second.